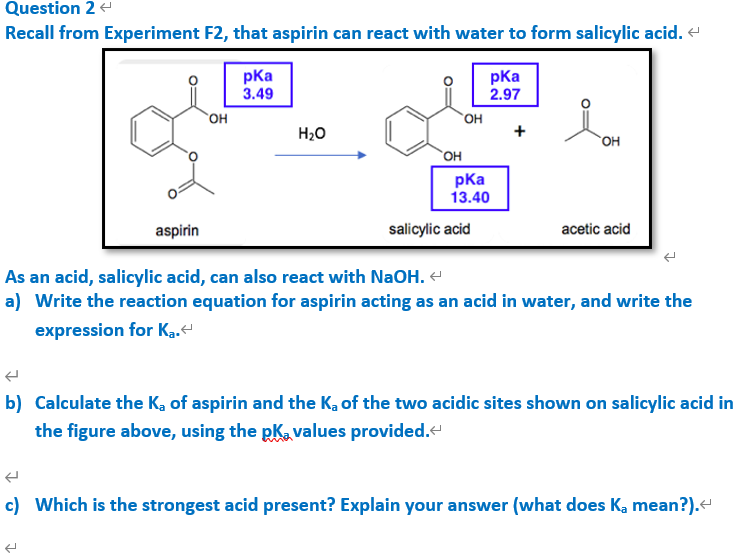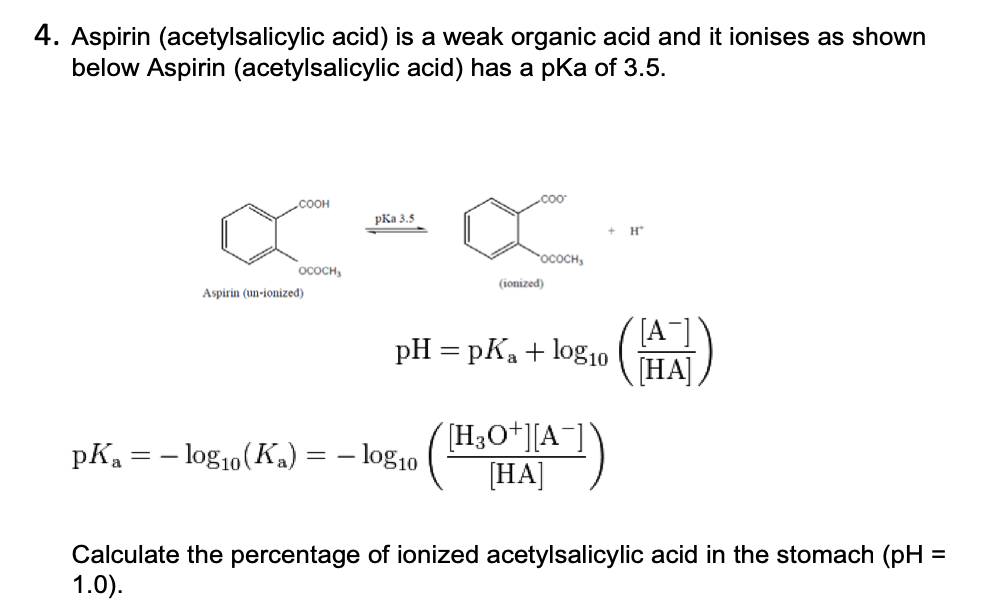
The ${\\text{pKa}}$ of acetyl salicylic acid (aspirin) is $3.5$ . The ${\\text{pH}}$ of gastric juice in the human stomach is about $2 - 3$ and the ${\\text{pH}}$ in the small intestine is

Ejercicio de p H para practicar la materia de farmacología - Ejercicio de pH La aspirina (ácido - Studocu
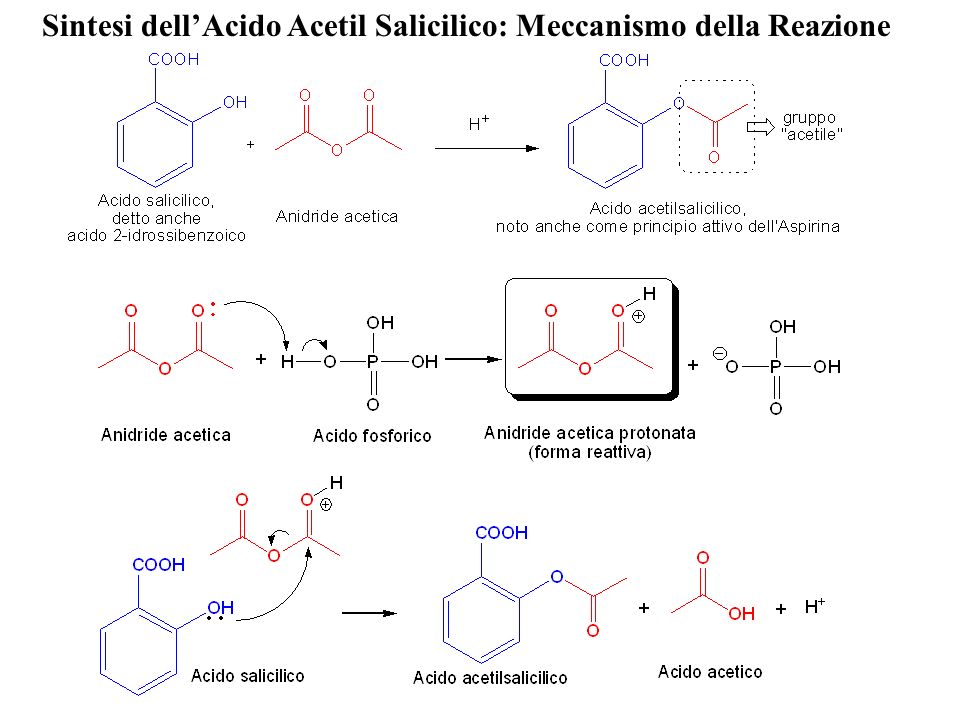
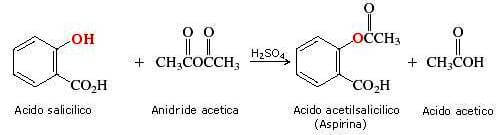
Esercitazione 3: Sintesi dell'Acido Acetil Salicilico Francesco De Riccardis Felix Hoffmann (chimica della Bayer) sintetizza l'acido acetilsalicilico (meglio. - ppt scaricare
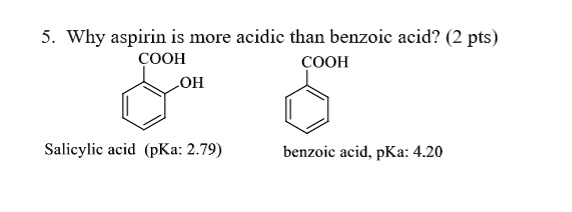
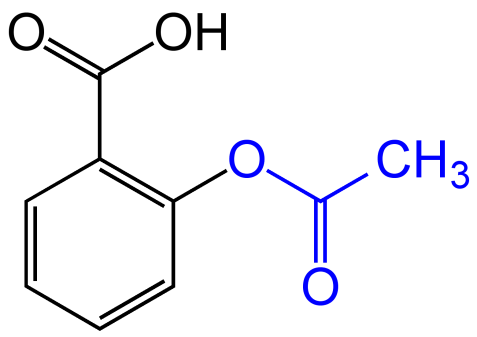
SOLVED: Why aspirin is more acidic than benzoic acid? pts) COOH COOH OH Salicylic acid (pKa: 2.79) benzoic acid. pKa: 4.20